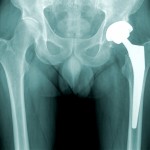
A story in the Asbury Park Press notes that it’s the latest in a series of about 40 product recalls announced by Johnson & Johnson since 2009.
Among them is the 2010 recall of an all-metal hip implant manufactured ...
continue reading...
A story in the Asbury Park Press notes that it’s the latest in a series of about 40 product recalls announced by Johnson & Johnson since 2009.
Among them is the 2010 recall of an all-metal hip implant manufactured ...
continue reading...
According to the article, flawed oversight of medical devices – essentially any medical product that doesn’t work through a chemical process – is an international problem that exists in the United States and Europe, as well as Canada.
The article specifically mentions two medical devices that serve ...
continue reading...
The implants in question, such as those manufactured by Johnson & Johnson’s DePuy Orthopaedics subsidiary, have both a ball and a socket coated in a ...
continue reading...
The all-metal implants feature both a ball and a socket coated in a combination of cobalt and chromium. They were originally marketed as more durable than hip replacements that feature ceramic or plastic. But industry-wide, manufacturers have been phasing out the ...
continue reading...
Newspaper The Australian reports that there is currently no requirement in Australia that sponsors of medical devices, which are companies that don’t produce the devices but supply them for sale, have insurance in case of problems with a device. The story says at least one senator intends to introduce legislation that would all sponsors ...
continue reading...
The story picks out Johnson & Johnson as an example of a company falling back on a new branding campaign in an effort to win back customers who have turned away because of recent problems.
In Johnson & Johnson’s case, those problems included a string of recalls, including both over-the-counter medicines and ...
continue reading...California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
