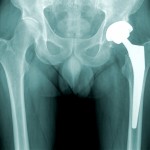
The all-metal implants feature both a ball and a socket coated in a combination of cobalt and chromium. They were originally marketed as more durable than hip replacements that feature ceramic or plastic. But industry-wide, manufacturers have been phasing out the all-metal models following widespread reports of the devices breaking down and needing replacement after only a few years, and shedding toxic metal debris in patients’ bodies.
In 2010, DePuy recalled a model of all-metal implant called the Articular Surface Replacement, or ASR. But the company continued to sell an all-metal version of a hip implant called the Pinnacle.
Meanwhile, DePuy found itself facing about 10,000 lawsuits from patients injured by one of the company’s recalled models. Lawyers for the plaintiffs have produced internal documents indicating that company officials were aware of potential hazards from the implants years before initiating the recall.
In the first of those cases to go to trial, a Los Angeles jury recently awarded $8.3 million in damages to one of the ASR implant recipients.
DePuy has not acknowledged any problems with the Pinnacle implants, but claims its discontinuation is due to declining sales and a change in U.S. Food and Drug Administration policy. The implants were originally approved through a FDA loophole that allows medical devices to go on the market without clinical testing in humans. But following the complaints about the all-metal devices, the FDA has announced that such testing will now be necessary.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a Johnson & Johnson product. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.nytimes.com/2013/05/17/business/jj-is-phasing-out-metal-replacement-hips.html?_r=0
