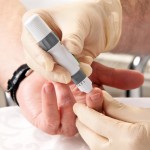
The story says Avandia was once the world’s top-selling diabetes drug. But in 2007, an analysis of other studies suggested that it raised the risk of heart attack by 43 percent. European regulators removed it from the market in 2010 and the ...
continue reading...
 Ranbaxy, a subsidiary of Japanese pharmaceutical company Daiichi Sankyo, has pleaded guilty to drug safety violations. According to a story in the New York Times, the company will pay $500 million in fines over allegations that it sold subpar drugs.
Ranbaxy, a subsidiary of Japanese pharmaceutical company Daiichi Sankyo, has pleaded guilty to drug safety violations. According to a story in the New York Times, the company will pay $500 million in fines over allegations that it sold subpar drugs.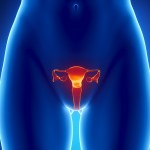 Reuters reports on a recently approved intrauterine device from Bayer, which is the first new device of its type that the FDA has approved in 12 years.
Reuters reports on a recently approved intrauterine device from Bayer, which is the first new device of its type that the FDA has approved in 12 years. The prestigious medical journal BMJ has published a damning investigative article on a crop of new diabetes drugs called “incretin therapies,” which include Byetta and Januvia.
The prestigious medical journal BMJ has published a damning investigative article on a crop of new diabetes drugs called “incretin therapies,” which include Byetta and Januvia. According to British newspaper the Mansfield and Ashfield Chad, a 26-year-old man left disabled from birth because of a traumatic brain injury has successfully sued for damages.
According to British newspaper the Mansfield and Ashfield Chad, a 26-year-old man left disabled from birth because of a traumatic brain injury has successfully sued for damages.