
All Eyes on California as $50 Billion Opioid Trial Sets Stage for What’s to Come

First Trial Scheduled in Opioid MDL

U.S. District Judge Daniel Polster had, to this point, sought ...
continue reading...Teva Suspends Zecuity Sales
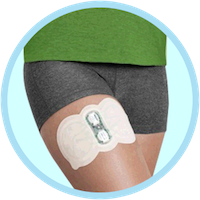
Zecuity’s launch has gone far worse than expected for Teva. The FDA issued a safety alert regarding the migraine relief patch earlier this month after a “large number of patients” experienced burns and scarring on the patch site.
Zecuity was intended ...
continue reading...Migraine Patch May Cause Severe Burns and Scarring

Various prescription pain relievers are available to ...
continue reading...In an annual report filed with the Securities and Exchange Commission, Teva Pharmaceutical Industries announced that the Department of Justice subpoenaed information relating to two of the company’s drugs. Copaxone, a drug to treat multiple sclerosis, is Teva’s top-selling drug. It is an injectable drug that the FDA approved for reducing the frequency of relapses, although there are claims that it may also reduce the progression of the disability. Azilect is used for ...
continue reading...