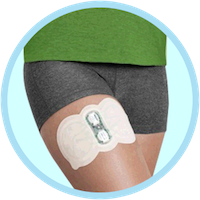
Zecuity’s launch has gone far worse than expected for Teva. The FDA issued a safety alert regarding the migraine relief patch earlier this month after a “large number of patients” experienced burns and scarring on the patch site.
Zecuity was intended to work by delivering a dose of sumatriptan to the patient through the skin. The battery-powered patch is wrapped around the arm or thigh. Unlike many other transdermal delivery systems, Zecuity can only be left in place for four hours at a time.
Shortly after launch, the FDA began receiving reports of severe skin reactions to the patch. These included redness, pain, discoloration, blistering, and cracks in the skin. Patients were advised to remove Zecuity at the first sign of trouble and were also warned against getting Zecuity wet in any way. This includes bathing and showering as well as swimming.
As a safety alert, the agency’s action stopped short of forcing a recall or ordering any other immediate action on the part of the manufacturer. Instead, the FDA’s initial action on Zecuity was strictly advisory. Doctors were encouraged to take special care to notify their patients of the potential for adverse reactions and to report those reactions via the Medwatch safety program.
Now that Teva has suspended all activity surrounding Zecuity, doctors are advised to stop any further use of the device, including advising patients not to use any remaining prescribed patches. This is, of course, very bad news for anyone who was hoping that Zecuity might provide them the relief they so desperately need from their migraines.
Up to 36 million Americans suffer from the debilitating headaches and anyone who has ever had one can tell you that they can redefine a patient’s interpretation of pain. Aside from being excruciating, migraine sufferers can also experience intense sensitivity to light, as well as nausea and vomiting. Chronic sufferers can endure this experience anywhere from 10 to 14 times a month.
Whether Zecuity will re-enter the marketplace is anyone’s guess at this point. For now, however, those whose hopes for relief were bound to a new pain relief delivery mechanism will have to return to more standard migraine relief strategies.
