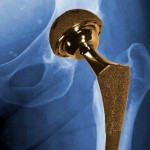
According to the article, prescription drugs have unique codes that can be used to track problems. But medical devices have no such identifiers and the FDA doesn’t even know how many devices are implanted ...
continue reading...
According to the article, prescription drugs have unique codes that can be used to track problems. But medical devices have no such identifiers and the FDA doesn’t even know how many devices are implanted ...
continue reading...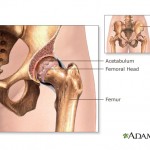
The federal court decision was in response to allegations that the company used misleading marketing for its antipsychotic drug Risperdal. The jury found that the company failed to properly disclose the drug’s possible side effects, like weight gain, ...
continue reading...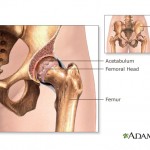
Government prosecutors charge that Johnson & Johnson paid kickbacks to Omnicare, the nation’s largest nursing home pharmacy, to get Omnicare to pick up the antipsychotic medication Risperdal and other Johnson & Johnson drugs.
The federal government ...
continue reading...Lopez McHugh has learned that transvaginal mesh devices, which are causing women extreme pain due to mesh erosion, extrusion, and failure, was approved by the FDA because they were similar another device on the market that has been recalled due to problems.
Yet this approval process is not limited to transvaginal mesh devices. A number of consumer advocacy groups have criticized the U.S. Food and Drug Administration’s 510(k) approval process, ...
continue reading...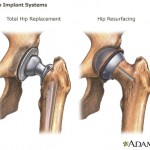
According to the Washington Post, shareholders were “upset over the health giant’s battered image amid unprecedented product recalls and ethical lapses.”
Weldon told them that the company started growing again last year after two years of sales declines, with revenue climbing ...
continue reading...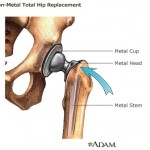
The article states that the rate of recalls, faulty devices, and repeat surgeries is increasing in an industry worth over $100 billion per year.
The first two devices the articles spotlights are ...
continue reading...California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
