The article states that the rate of recalls, faulty devices, and repeat surgeries is increasing in an industry worth over $100 billion per year.
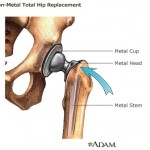
The first two devices the articles spotlights are DePuy Orthopaedics artificial hip implants, and vaginal mesh implants.
Both were approved through the FDA’s 510(k) process, which allows devices to be approved without clinical data, based on their alleged similarity to previously approved devices.
According to Scientific American, testing on DePuy’s all-metal hip implants began once they went to market. The article cites the British Hip Society, which said the failure rate of the DePuy implant was so high that almost half of all patients who got the implant required additional surgery within six years to correct problems.
The story is similar with transvaginal mesh. The FDA first approved a mesh device to treat pelvic organ prolapse and urinary incontinence in the late 1990s, based on its alleged similarity to a hernia treatment.
The device’s original manufacturer voluntarily recalled it in 1999 after numerous complaints of pain, infections, and injuries. But that earlier approval nonetheless served as the basis for approval of Johnson & Johnson’s surgical mesh product. Since then, thousands of women who received the device have filed lawsuits over complications, the most common of which concern the mesh eroding and protruding through the walls of the bladder or vagina.
See the article here: https://www.scientificamerican.com/article.cfm?id=four-medical-implants-escaped-fda-scrutiny
