Jurors in Louisiana found Johnson & Johnson officials violated the state’s Medicaid fraud laws 35,542 times over Risperdal. Lawyers for the state argued that the company defrauded Louisiana’s Medicaid program by wrongfully marketing the drug to doctors as safer and better than competing medicines so they could sell it at an artificially inflated price.
A number of other ongoing cases in state and federal courts allege that the company improperly marketed dangerous medical products.
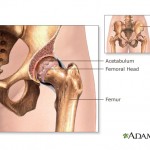
The U.S. Food and Drug Administration is investigating metal-on-metal hip implants manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics. Studies show that nearly half of the patients who receive the devices need additional surgery within six years because they fail prematurely. In many cases, the implants also leave toxic metal debris in patients’ bodies.
Although the hip implants were recalled in 2010, an investigation by British medical journal The Lancet found internal company documents indicating that Johnson & Johnson officials were aware as early as 2005 of the device’s high early failure rate, yet continued to market it.
The company is also facing legal action over its transvaginal mesh implants, used to treat pelvic organ prolapse and urinary incontinence. Up to 10 percent of the devices fail within a year, causing health problems including debilitating pain and infection.
Although Johnson & Johnson’s Ethicon unit recently announced it would stop selling four lines of vaginal mesh devices, the company marketed them despite thousands of reports of device failure, and despite an FDA order to stop.
The company also faces additional legal action over Risperdal. The Louisiana ruling came a day after Johnson & Johnson agreed to pay $181 million to resolve claims by 36 states that they improperly marketed and advertised both Risperdal and Invega, another antipsychotic medicine.
In April, a judge in Arkansas ordered the drugmaker to pay $1.2 billion in fines over Risperdal marketing. Three months earlier, Johnson & Johnson decided to end a trial in Texas over the drug’s sales with a $158 million settlement. And in June 2011, a judge in South Carolina ordered Johnson & Johnson to pay $327 million in penalties for deceptively marketing the medicine.
If you’ve received a DePuy hip or transvaginal mesh implant, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries from either device, you should also consult with a lawyer familiar with the DePuy hip implant or transvaginal mesh case to discuss your legal rights.
See the story here:
https://www.nj.com/business/index.ssf/2012/08/johnson_johnson_loses_bid_to_t.html
