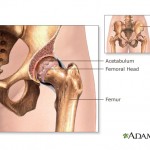
According to U.S. News and World Report, the 6-to-4 decision came as a surprise because an FDA briefing document filed Monday in advance of the ruling gave the drug a favorable assessment.
But panel members reportedly said company studies did not provide enough ...
continue reading...
 In a Motley Fool article for MSNBC, Sara Wright recommends against investing in manufacturers of dangerous medical devices that made it onto the market through a regulatory loophole.
In a Motley Fool article for MSNBC, Sara Wright recommends against investing in manufacturers of dangerous medical devices that made it onto the market through a regulatory loophole.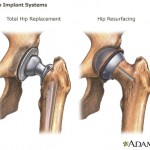 Consumers Union, the policy and advocacy arm of Consumer Reports, has placed a full-page print ad in Washington, D.C.-based publication Politico, calling on Congress to close a loophole that allows dangerous devices to make it on the market without clinical testing.
Consumers Union, the policy and advocacy arm of Consumer Reports, has placed a full-page print ad in Washington, D.C.-based publication Politico, calling on Congress to close a loophole that allows dangerous devices to make it on the market without clinical testing.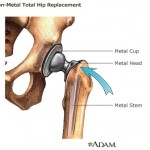 Lawyers representing Pennsylvania and Johnson & Johnson both argued their side before a panel of seven Commonwealth Court justices on Wednesday, in a case involving allegations that Johnson & Johnson defrauded Pennsylvania out of millions of taxpayer dollars.
Lawyers representing Pennsylvania and Johnson & Johnson both argued their side before a panel of seven Commonwealth Court justices on Wednesday, in a case involving allegations that Johnson & Johnson defrauded Pennsylvania out of millions of taxpayer dollars.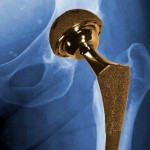 Commonwealth Court judges are scheduled to hear an appeal of the decision dismissing Pennsylvania’s 2008 lawsuit against Johnson & Johnson. The company is accused of fraudulently profiting from sales of the antipsychotic drug Risperdal through the Medicaid program.
Commonwealth Court judges are scheduled to hear an appeal of the decision dismissing Pennsylvania’s 2008 lawsuit against Johnson & Johnson. The company is accused of fraudulently profiting from sales of the antipsychotic drug Risperdal through the Medicaid program.