The plaintiff claims he suffered metal poisoning and other health problems from DePuy’s ASR hip replacement device, which he had implanted in 2007. Johnson & Johnson recalled about 93,000 of ...
continue reading...
The plaintiff claims he suffered metal poisoning and other health problems from DePuy’s ASR hip replacement device, which he had implanted in 2007. Johnson & Johnson recalled about 93,000 of ...
continue reading...
The ASR artificial hip implant, manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics, has been the basis of about 10,000 lawsuits based on their tendency to break down early and leave toxic metal debris in patients’ bodies.
And at ...
continue reading...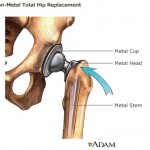
Johnson & Johnson revealed the government investigation in a recent report to the Securities and Exchange Commission.
According to the regulatory filing, the U.S. Attorney’s Office in Massachusetts and the Civil Division of ...
continue reading...
The company is already facing about 10,000 lawsuits connected to another model of all-metal hip implant called the ASR, which was recalled in 2010 over its tendency to break down early and leave toxic metal debris in patients’ bodies.
The Reuters story ...
continue reading...
Studies show that nearly half of the implants, made by Johnson & Johnson subsidiary DePuy Orthopaedics, break down and need replacement after only a few years. They also have a tendency to ...
continue reading...
“Though the company says the evidence will ultimately show that it acted appropriately, it clearly has a lot of explaining to do,” the editorial says.
The model in question ...
continue reading...California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
