The priority review status is granted by the FDA to expedite the review of drugs to treat serious diseases and fill unmet medical needs. It means the drug will be reviewed ...
continue reading...
The priority review status is granted by the FDA to expedite the review of drugs to treat serious diseases and fill unmet medical needs. It means the drug will be reviewed ...
continue reading...
In a press release, CSPI says that Splenda Essentials’ marketing is designed to give the false impression that the sweetener confers health benefits, such as helping its users lose weight and avoid disease.
The lawsuit ...
continue reading...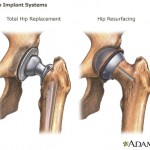
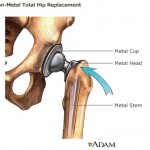
According to the Wall Street Journal, the U.S. Justice Department sent J&J’s Janssen Pharmaceuticals unit a letter in April, requesting documents related to the marketing and promotion of the antibiotic Doribax.
In ...
continue reading...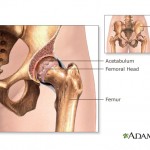
The report notes that Warren Buffett’s Berkshire Hathaway Inc. dropped two-thirds of its holdings in the company, following a $1.1 billion fine from an Arkansas jury that ruled the firm misled doctors and patients about the risks of antipsychotic medication Risperdal.
Johnson & Johnson ...
continue reading...
Federal and state investigators have been examining the company’s Risperdal marketing practices for almost a decade, according to the Inquirer. The $181 million agreement is separate from a continuing federal investigation that could lead to criminal charges against Johnson ...
continue reading...
Reuters reports that Synthes issued the recall on July 5, citing the potential for the putty to catch fire if it came in contact with electrosurgical cautery systems during ...
continue reading...California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
