
The story characterizes it as “a bit of bright news” from Ethicon, noting that the company has initiated a number of recalls lately for items including hemorrhoidal staples and wound products.
And Ethicon is currently on trial ...
continue reading...
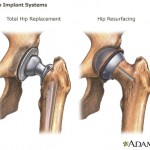 A piece in The Economist ironically describes Johnson & Johnson as the “market leader” for publicity problems, based on the company’s disastrous series of product recalls in recent years.
A piece in The Economist ironically describes Johnson & Johnson as the “market leader” for publicity problems, based on the company’s disastrous series of product recalls in recent years. The U.S. Food and Drug Administration has sent a warning letter to medical device manufacturer St. Jude Medical, warning of problems with manufacturing and quality control at the California plant where the company makes implantable heart defibrillators.
The U.S. Food and Drug Administration has sent a warning letter to medical device manufacturer St. Jude Medical, warning of problems with manufacturing and quality control at the California plant where the company makes implantable heart defibrillators.