The report says many states have recovered just as much, if not more, money from pharmaceutical companies accused of defrauding Medicaid programs as they spent on all Medicaid fraud enforcement. A total of $6.6 billion had been recovered from the beginning of 2012 through mid-July by both the federal government and states, which is a record amount.
Three companies – GlaxoSmithKline, Johnson & Johnson and Abbott – were responsible for two-thirds of the financial penalties paid out to the federal and state governments during a study period that went from Nov. 2, 2010, to July 18, 2012.
Though the dollar amounts may seem large, Public Citizen points out that they are still much too low to deter future violations. A new release from the organization states: “The $30 billion paid out by pharmaceutical companies in settlements to the federal government and states since 1991 represents just a little more than two-thirds of the profits made by the 10 largest drug companies in 2010 alone.”
Some of those companies have a history of complaints by Public Citizen and other advocacy groups.
For example, Johnson & Johnson has been the subject of thousands of lawsuits, alleging that company officials suppressed information indicating that their medical devices harmed patients who had them implanted.
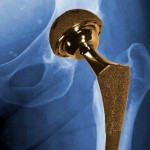
Johnson & Johnson’s DePuy Orthopaedics division recalled a line of all-metal hip implants in 2010 after nearly half of the patients who received them had to get them replaced within six years. Patients who received the implants also had problems with toxic metal debris breaking off and getting in their bodies.
And Johnson & Johnson recently recalled four different types of transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, following widespread complaints that they caused severe pain and injuries for recipients.
Lawyers for the plaintiffs have produced evidence indicating that the company marketed both devices for years after company officials learned of the health hazards they presented.
Public Citizens says that overcharging health programs, mainly in the form of drug pricing fraud against state Medicaid programs, was the most common violation during the study period. The unlawful promotion of drugs was associated with the largest penalties, as in the prior report.
A Public Citizen news release quotes Dr. Sammy Almashat, a researcher with Public Citizen’s Health Research Group and the study’s author, as saying: “It should come as no surprise that states facing Medicaid budget shortfalls are finally deciding to root out fraud that likely has cost their taxpayers billions of dollars over the years. What this new report unequivocally shows is that those states that have chosen to hold the pharmaceutical industry accountable have largely seen their enforcement efforts pay for themselves.”
Patients should consult their doctors before making any changes in their medication. And you should consult with a doctor if you have any ongoing symptoms or health concerns from a transvaginal mesh or metal-on-metal hip implant. If you or a loved one have significant injuries from a medical implant or an SSRI, you should also consult with an SSRI, DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.nj.com/business/index.ssf/2012/09/johnson_johnson_other_drugmake.html
