
The Scientist cites an article in the Journal of the American Medical Association, which states that the FDA fast-tracked nearly half of all the 35 drugs it reviewed in 2011.
Among them was the blood thinner dabigatran, sold ...
continue reading...
The Scientist cites an article in the Journal of the American Medical Association, which states that the FDA fast-tracked nearly half of all the 35 drugs it reviewed in 2011.
Among them was the blood thinner dabigatran, sold ...
continue reading...
According to the report, the mesh devices have caused “life-changing harm” for a significant number of the women who received them because of their tendency to erode in patients’ bodies.
The implants are sling-like devices designed to treat urinary incontinence and pelvic organ prolapse, which ...
continue reading...
The proposals come in the wake of a global scandal in which hundreds of thousands of women around the world received breast implants prone to rupture and leaking, made by defunct French company Poly Implant Prothese.
Critics say that because of weak EU safety standards, regulators had failed to stop the implants for more ...
continue reading...
According to a Reuters report, the biggest recalls can lead to billions of dollars in expenses, and medical device makers are already worried about weaker global demand for many of their products and the impact of a new U.S. tax on ...
continue reading...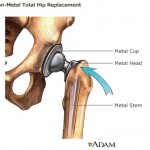
Terms of the settlement weren’t disclosed, Bloomberg reports.
The lawsuits are among more than 400 in which J&J and its Janssen unit were accused of injuries caused by Risperdal. More than 100 of those suits involve claims that the drug caused young ...
continue reading...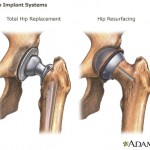
The letter was sent following an agency inspection of the company’s plant in Puerto Rico, which found manufacturing and testing issues related to the company’s Trilogy Acetabular System products — implanted in hip-replacement surgery.
The report came ...
continue reading...California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
California
(877) 737-8525 – Toll Free
(949) 737-1501
(949) 737-1504 – Fax
New Jersey
(856) 273-8500
(856) 273-8502 – Fax
Pennsylvania
(877) 703-7070 – Toll Free
(215) 952-6910
(215) 952-6914 – Fax
