A New York Times report quotes Steven Silverman, director of compliance for the U.S. Food and Drug Administration’s medical devices division, as saying: “Candidly, I just think it’s bad business and not ...
continue reading...
A New York Times report quotes Steven Silverman, director of compliance for the U.S. Food and Drug Administration’s medical devices division, as saying: “Candidly, I just think it’s bad business and not ...
continue reading...
He previously had to make a long drive and arrive at a dialysis center at 5 a.m. in order to finish the 4-hour treatment, then make it to work on time. Doctors told him that routine would be necessary three ...
continue reading...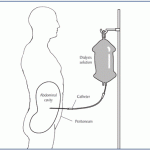
Researchers have concluded that fluid in the lungs is a better predictor of the risk of dying or having a heart attack than other symptoms of heart failure.
Patients undergoing kidney dialysis are at risk of heart failure. In some cases, the dialysis itself may be ...
continue reading...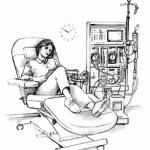
The agency’s warning letter alleges that Fresenius failed to adequately test artificial kidneys — called “dialyzers” – manufactured in Utah, to make sure they adequately meet design specifications.
The FDA stopped short of issuing a recall in this case. But the ...
continue reading...