A Reuters story says that payment is part of a $1.2 billion settlement the company agreed to pay Arkansas in 2012, based on the state’s claims that Johnson & Johnson downplayed the risks of the antipsychotic drug Risperdal.
Arkansas alleged that Johnson & Johnson deceived thousands of doctors by marketing Risperdal for unapproved uses in children and the elderly, which led to the state’s Medicaid insurance greatly overpaying for the drug.
Johnson & Johnson is currently defending itself in other trials alleging deception on the company’s part.
In a New Jersey trial, a woman claims she was injured by a transvaginal mesh implant that Johnson & Johnson subsidiary DePuy Orthopaedics manufactured. Hers is the first of about 6,000 cases to go to trial, from patients who allege the implants caused problems including chronic pain, infection and organ perforation.
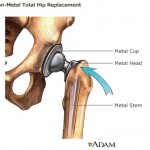
And in Los Angeles, the recipient of an all-metal hip implant manufactured by Johnson & Johnson’s DePuy Orthopaedics division is suing over allegations that toxic debris from the device caused metal poisoning.
His is the first of about 10,000 cases from patients claiming that similar hip implants either left toxic metal debris in their bodies, or broke down and needed replacement after only a few years.
In both cases, plaintiffs claim the company was aware of problems with the devices for years, even as it allowed them to be sold.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a Johnson & Johnson product. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.reuters.com/article/2013/02/01/usa-baileyperrinfee-idUSL1N0B1JMH20130201
