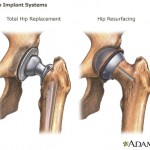
The letter was sent following an agency inspection of the company’s plant in Puerto Rico, which found manufacturing and testing issues related to the company’s Trilogy Acetabular System products — implanted in hip-replacement surgery.
The report came ...
continue reading...
 Researchers in the U.K. have recommended that women not receive all-metal hip replacements because of the procedure’s early failure rate, Bloomberg reports.
Researchers in the U.K. have recommended that women not receive all-metal hip replacements because of the procedure’s early failure rate, Bloomberg reports. Urinary incontinence can be an embarrassing condition that often involves some inconvenient lifestyle changes.
Urinary incontinence can be an embarrassing condition that often involves some inconvenient lifestyle changes. Consumer Reports conducted a survey about antidepressant use, compiling answers from 1,500 respondents. According to an article accompanying the survey results, the findings contradict the implications of antidepressant advertisements, which imply that pills are the only answer for serious depression or anxiety disorders and that they work for everyone.
Consumer Reports conducted a survey about antidepressant use, compiling answers from 1,500 respondents. According to an article accompanying the survey results, the findings contradict the implications of antidepressant advertisements, which imply that pills are the only answer for serious depression or anxiety disorders and that they work for everyone. A story in the Tampa Bay Times warns readers that a long airplane trip can put them at risk of suffering a blood clot.
A story in the Tampa Bay Times warns readers that a long airplane trip can put them at risk of suffering a blood clot.