According to the Washington Post, shareholders were “upset over the health giant’s battered image amid unprecedented product recalls and ethical lapses.”
Weldon told them that the company started growing again last year after two years of sales declines, with revenue climbing 5.6 percent to $65 billion.
Weldon and Gorsky also said the company has instituted new quality procedures in the wake of about 30 recalls of medical devices and prescription drugs. The causes for those recalls, according to the report, included nauseating packaging odors, wrong levels of active ingredients in pills, and glass and metal shards in liquid medicines.
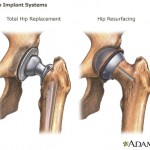
Another high-profile recall is the all-metal hip implant manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics, pulled from the market in 2010. Complaints about the implant include a failure rate requiring almost half of all recipients to get a replacement within six years, and metal toxic metal debris breaking off in patients’ bodies.
Another Johnson & Johnson surgical implant, transvaginal mesh used to treat urinary incontinence and pelvic organ prolapse, has prompted thousands of lawsuits over its high early failure rate.
See the report here:
https://www.washingtonpost.com/business/johnson-and-johnsons-incoming-outgoing-ceos-reassure-shareholders-on-recalls-other-problems/2012/04/26/gIQAxHkPjT_story.html
