Symptoms include:
- Shoulder pain
- Stiffness
- Limited range of motion
- Popping or grinding when the shoulder is in motion.
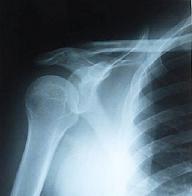
A diagnosis can be made by x-rays showing the narrowing of the shoulder joint space.
Manufacturers Failed to Warn of Pain Pump Injuries
PAGCL is a debilitating condition that typically results in permanent shoulder pain. There is no treatment except for arthroplasty, which is the replacement of the shoulder joint (an expensive and painful procedure).
It has been alleged that the manufacturers of the pain management pump failed to warn the medical community that the devices could cause permanent injury. Manufacturers include Stryker Corp., I-Flow Inc., DJ Orthopedics Inc., Sorenson Medical Products Inc., Moog Inc., Curlin Medical Inc., McKinley Medical LLC, Breg Inc., and Sgarlato Labs.
Lopez McHugh is currently investigating incidents of PAGCL due to defective pain pumps, and is representing consumers across the country. If you or someone you know has had complications and is suffering from defective pain pump, contact our attorneys to learn about your legal rights.
