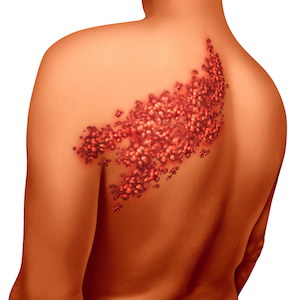
Zostavax is recommended by the CDC for use in people over the age of 60. Shingles and chickenpox are caused by the same virus, and Merck emphasizes that fact in its advertising for Zostavax. With the promise that, “if you had chickenpox, the shingles virus is already inside you,” Merck asserts that one in three people who dealt with chickenpox will contract shingles in their lifetime.
Interestingly, Zostavax itself seems to offer little assistance in preventing this eventuality. Studies indicate an effectiveness rate of just 51% for a drug referred to as a vaccine; the same term used for the medications responsible for actually wiping out dangerous conditions like polio and smallpox. What’s more, Zostavax may actually cause shingles rather than prevent it.
The desire to prevent shingles comes from much more than a patient’s desire to avoid an incredibly painful skin condition. Shingles comes with a frightening list of possible side effects and complications that includes meningitis, encephalitis, stroke, and congestive heart failure. Evidence suggests that adverse reactions to Zostavax might be directly responsible for 90 deaths across the United States.
Up until recently, Zostavax’s warning label did not contain a statement explicitly linking the vaccine to the condition it is intended to prevent. That changed earlier this month, however, when the language was updated to warn of the possibility of Zostavax causing shingles in “individuals who are immunodeficient or immunosuppressed.”
