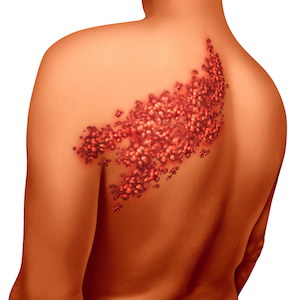
The previous label made no direct mention of the fact that, if taken by someone who has a suppressed or inactive immune system, Zostavax could actually cause shingles in the vaccinated patient. This is an important point, as the patient would be administered the vaccine specifically to avoid the painful – and potentially life-threatening – condition.
As previously written, “ZOSTAVAX,” the label read, “is a live, attenuated varicella-zoster vaccine and administration may result in disseminated disease in individuals who are immunosuppressed or immunodeficient.” Without an explicit statement, “disseminated disease” could mean any number of conditions; either related or unrelated to the shingles virus.
The new label is much more direct in its instruction and meaning. “Do not administer ZOSTAVAX to individuals who are immunodeficient or immunosuppressed due to disease or therapy,” it says, “as serious or fatal disseminated vaccine strain varicella-zoster virus disease may occur.”
The label update is the latest news surrounding a drug that continues to face increasing levels of scrutiny. Targeted at adults aged 50 years or older, the CDC actually recommends that Americans over the age of 60 get the shingles prevention vaccine. Television viewers are all too familiar with the scary sounding voice warning them that if they’d had chicken pox as a child, that the shingles virus is “already inside them.”
What the commercial fails to mention is that Zostavax appears to only be about 51% effective and may be responsible for the deaths of close to 100 people across the country. Over 1,100 adverse events have been recorded in the Vaccine Adverse Events Reporting System. 90 of those patients eventually died from complications directly related to the Zostavax vaccine.
Numerous Zostavax lawsuits have been filed across the country as a result of these and other concerns.
