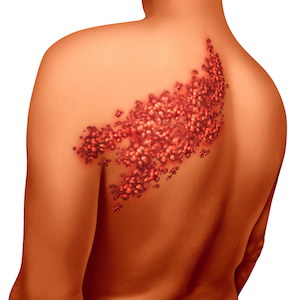
Zostavax is targeted at people aged 50 years or older and the CDC has gone so far as to issue a formal recommendation for everyone over the age of 60 to receive the vaccine. Zostavax contains live varicella zoster virus – the same infection that causes both chickenpox and shingles. Those who had chickenpox as a child still have the virus living within them. The idea is to stimulate and condition the body’s immune system to continue to be on the lookout for varicella zoster as the patient ages. This could help to prevent a shingles flare-up later in life.
“Could” is, unfortunately the operative word in this case, as Zostavax is only shown to be about 51% effective at doing its job. And, if that wasn’t bad enough, there is significant evidence to suggest that Zostavax may actually cause the condition it seeks to prevent – exposing patients to a painful and life-altering skin condition they might not have otherwise had to contend with.
The damage caused by shingles can go far past the painful skin rash the condition initially presents as. Side effects of chickenpox and shingles can also include meningitis, encephalitis, stroke, and congestive heart failure.
Zostavax may also be directly responsible for 90 deaths across the United States. An analysis of adverse event reporting data found in the Vaccine Adverse Events Reporting System from 1990 to September of 2015 linked over 1,100 events specifically to Vostavax. 90 of those patients eventually died from their complications.
In response to this latest Zostavax lawsuit, a Merck representative states that the company “stands behind the demonstrated safety and efficacy” of Zostavax, noting that over 36 million doses of the shingles vaccine have been distributed since 2006.
For those 36 million people, a 51% effectiveness rate and a lawsuit alleging that the company that made the injection “knew, or should have known, that its product caused viral infection, and was therefore not safe for administration to consumers” does not bode well.
