In 2010, the company’s McNeil Consumer Healthcare unit recalled more than 280 million packages of over-the-counter medications, which included Motrin, children’s Tylenol liquid and Benadryl.
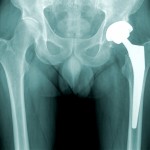
Also in 2010, Johnson & Johnson’s DePuy Orthopedics unit recalled two popular all-metal artificial hip replacement models, which have subsequently been the basis of about 10,000 lawsuits. A jury recently awarded $8.3 million to a man injured by one of those implants, which have a tendency to break down and require replacement after only a few years, and to shed toxic metal debris in patients’ bodies.
In another high-profile case involving a Johnson & Johnson medical device, a jury awarded $11.1 million to a woman injured by a vaginal mesh implant from the company’s Ethicon division. That case represented the first of more than 11,000 lawsuits about the devices to go to trial, filed by women complaining of health problems that include infection, organ perforation and chronic pain.
CEO Alex Gorsky recently succeeded Bill Weldon largely as a result of those complaints. According to the story, Gorsky tried to reassure the shareholders that the company is making progress in improving quality standards and efficiency.
Gorsky told the shareholders that Johnson & Johnson posted an adjusted profit of $4.1 billion in the first quarter, and has produced 29 consecutive years of increased earnings.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a Johnson & Johnson product. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://articles.washingtonpost.com/2013-04-25/business/38810776_1_jamp-argyle-country-club-corrie-tayman
