“Manufacturing problems and recalls are not just embarrassing,” she writes, “but they are a worrisome sign that management has taken its eye off the ball in many key respects.”
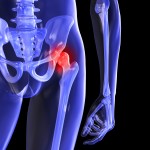
Maller specifically mentions a recall that Johnson & Johnson initiated for its LPS Diaphyseal Sleeve, a device used in reconstructive knee surgery that the company’s DePuy Orthopaedics division manufactured.
The FDA said the recall was necessary because of the device’s potential for fractures.
The company has gone through other financial and public relations troubles because of its implants.
In the first of about 11,000 lawsuits to go to trial, a New Jersey jury awarded a woman $11.1 million to compensate her for injuries from a vaginal mesh implant that Johnson & Johnson subsidiary Ethicon manufactured. Common complaints from the mesh implants include chronic pain, infection and organ perforation.
And a Los Angeles jury has awarded $8.3 million in damages in the first of about 10,000 lawsuits involving ASR hip implants from DePuy. A number of studies show nearly half of the implants break down and need replacement within six years. They also tend to shed toxic metal debris in patients’ bodies.
Particularly worrying from an investor’s perspective, Maller writes, is that the company’s board recently gave CEO Bill Weldon a 3 percent raise, after initially pledging to cut his bonus by half. She notes that the raise came after one of the company’s worst years in recent memory, and questions why he would deserve it.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a Johnson & Johnson product. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the piece here:
https://seekingalpha.com/article/1261841-johnson-johnson-throwing-in-the-towel
