According to Reuters, both American and European anti-trust regulators have long criticized the practice. It recently came to the forefront again after European Union officials accused Johnson & Johnson of making a pay for delay arrangement with Novartis, which blocked the introduction of a generic version of the painkiller fentanyl.
Johnson & Johnson has repeatedly faced legal trouble over allegations that it sacrifices the well-being of consumers for profits. Two ongoing trials center on charges that company knowingly sold dangerous medical devices without warning patients.
The plaintiff in a New Jersey trial alleges that one of Johnson & Johnson’s transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, forced her to quit her job because of chronic pain.
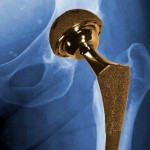
And the plaintiff at another trial in Los Angeles claims that his all-metal hip implant manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics needed to be replaced within only a few years, and left toxic metal debris in his body.
Both are the first cases to go to trial among thousands of similar complaints involving both devices. And in both cases, attorneys for the plaintiffs cite internal documents indicating that Johnson & Johnson officials knew in advance about problems with the products.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip or transvaginal mesh implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.reuters.com/article/2013/02/05/usa-pharmaceuticals-antitrust-idUSL1N0B5I9Q20130205
