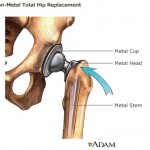
Metal-on-metal hip implants, which have both a ball and socket coated in an alloy made of cobalt and chromium, were originally introduced as an ostensibly more durable alternative to earlier plastic and ceramic models. But a number of studies have indicated that nearly half of them fail prematurely after only a few years.
Many patients who’ve received the all-metal devices have also experienced problems with toxic metal debris breaking off and getting into their soft tissues.
According to Bloomberg, the adverse events reported to the U.S. Food and Drug Administration almost quadrupled to 682 in 2008 from the year earlier.
After Johnson & Johnson’s DePuy Orthopaedics unit began recalling the devices in 2010, the number of adverse reports rose again — totaling 12,137 in 2010 for the metal-on-metal devices, compared with 6,332 associated with other types of hip implants.
The side effect reported most often was revision, meaning surgery to remove or replace the implant. Revisions for the metal hip joints totaled 14,131. The second most common side effect was pain, which accounted for 8,704 reports.
The report says all-metal hip implant use peaked in 2006 and 2007, at an estimated 30 percent of the market. It has now dropped to about 10 percent because of the adverse event reports.
Plaintiffs in thousands of lawsuits cite internal Johnson & Johnson documents indicating that company officials were aware of the problems associated with the implants for years before the recall, yet continued to market them anyway.
If you’ve received an all-metal hip implant, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries, you should also consult with a DePuy hip lawyer familiar with the case to discuss your legal rights.
