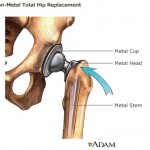
A report on Boston.com says the all-metal implants – in which both the ball and sockets are coated with metal – were originally introduced on the assumption that they would be safer and more durable than existing plastic and ceramic versions.
But in March, British experts at the world’s largest artificial joint registry told doctors to stop using the devices, because their analysis found that more than 6 percent of patients with metal hips needed them replaced after less than five years, compared with just 2 percent of people who had ceramic or plastic joints.
Hip replacements are supposed to last between 10 to 15 years, the report says.
British regulators now recommend that people with the implants get annual blood tests to ensure that no dangerous metals are seeping into their bodies as the components rub against each other.
The FDA has not made any recommendations for the estimated 500,000 American patients with the devices.
Unlike other countries, America has no national registry to track the artificial hips’ success or failure rate. But according to the report, the FDA received 16,800 negative event reports involving metal hips between 2000 and 2011.
And a registry set up by Minnesota’s HealthEast Care System recorded four times as many replacement surgeries for patients with metal-on-metal hips as those with other implant types.
If you’ve received an all-metal hip implant, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries, you should also consult with a DePuy hip lawyer familiar with the case to discuss your legal rights.
See the report here:
https://www.boston.com/business/healthcare/2012/06/26/fda-probes-safety-issues-with-metal-hip-implants/S5DL91jpgvDAntQqIcCtfN/story.html
