A jury in Arkansas ruled that Johnson & Johnson officials used deceptive tactics to market antipsychotic drug Risperdal, misleading doctors about the drug’s risks and improperly marketing it for unapproved uses, Bloomberg reports.
The decision in Arkansas marks the third such jury verdict against Johnson & Johnson over allegations that the pharmaceutical giant concealed Risperdal’s risks and tricked Medicaid regulators into paying more than they should have for the medication.
Juries also decided against the company in Louisiana and South Carolina.
A total of 11 states have sued Johnson & Johnson in an attempt to get reimbursement for Medicaid or other public funds paid on Risperdal prescriptions. The lawsuits allege that Johnson & Johnson promoted the drug for unapproved uses or downplayed risks.
A judge will now decide whether to fine the company over its actions in Arkansas. The state is seeking $1.25 billion in penalties.
The Bloomberg report quotes Arkansas Attorney General Dustin McDaniel as saying the company “lied to patients and doctors because they cared more about profits than people.”
Users of transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, have complained about health problems including organ perforation and debilitating pain.
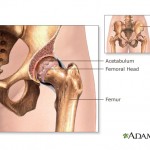
And metal-on-metal hip implants manufactured by a subsidiary of Johnson & Johnson are being investigated by the U.S. Food and Drug Administration after thousands of reports that they fail prematurely and leave toxic metal debris in patients’ bodies.
Plaintiffs claim the company actively marketed both devices despite being aware of those problems.
See the report here: https://www.bloomberg.com/news/2012-04-10/j-j-s-risperdal-marketing-violated-law-arkansas-jury-rules-1-.html?
