The final days of summer have come to a close and thoughts are turning to cooler temperatures, holidays, and end-of-year celebrations. This also means that we’re taking a few moments to reflect on a year of news and posts that show the worst of what can happen when corporations and governments – two entities that, in many ways, have become indistinguishable from one another – put finance, profit, and politics over public safety.
It’s been a year of progress on many ...
continue reading...
 What began as a $500 million verdict in five plaintiffs’ favors in 2016 and was later cut to $151 million has now been reduced to nothing. At the same time, Johnson & Johnson – once on the losing side of a significant decision in a bellwether trial over its DePuy division’s Pinnacle hip implants – can take a case out of the “loss” column for now.
What began as a $500 million verdict in five plaintiffs’ favors in 2016 and was later cut to $151 million has now been reduced to nothing. At the same time, Johnson & Johnson – once on the losing side of a significant decision in a bellwether trial over its DePuy division’s Pinnacle hip implants – can take a case out of the “loss” column for now. “You got to know when to hold ‘em. Know when to fold ‘em. Know when to walk away, and know when to run.”
“You got to know when to hold ‘em. Know when to fold ‘em. Know when to walk away, and know when to run.”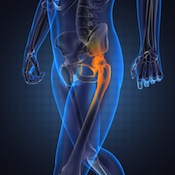 Another Wright hip implant lawsuit has joined the growing Georgia multidistrict litigation, this time alleging that
Another Wright hip implant lawsuit has joined the growing Georgia multidistrict litigation, this time alleging that  The U.S. Food and Drug Administration (FDA) has issued a
The U.S. Food and Drug Administration (FDA) has issued a