A Wall Street Journal report on the policy says that it would require the high-risk devices to carry identification numbers. Jeffrey Shuren, director of the FDA’s medical-device center, described the plan as “a major game-changer” and said the agency plans to ramp up efforts to identify malfunctioning medical devices early.
For a long time, public safety advocates ...
continue reading...
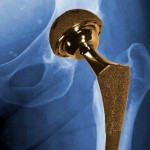
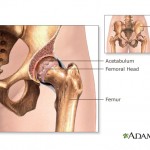 Members of a U.S. Food and Drug Administration panel that recently discussed metal-on-metal artificial hip implants said they wouldn’t recommend that patients get them, although the FDA as a whole has so far stopped short of an official ruling on the devices.
Members of a U.S. Food and Drug Administration panel that recently discussed metal-on-metal artificial hip implants said they wouldn’t recommend that patients get them, although the FDA as a whole has so far stopped short of an official ruling on the devices. The $3 billion fine recently leveled against GlaxoSmithKline is the largest settlement ever involving a pharmaceutical company. But in light of the enormous profits that the British pharmaceutical giant brings in, some critics wonder whether even that will be enough to dissuade companies from engaging in dangerous fraud.
The $3 billion fine recently leveled against GlaxoSmithKline is the largest settlement ever involving a pharmaceutical company. But in light of the enormous profits that the British pharmaceutical giant brings in, some critics wonder whether even that will be enough to dissuade companies from engaging in dangerous fraud. A higher price as a selling point may seem counterintuitive. Why would somebody purchase a product specifically because it costs more, particularly if safer alternatives are available?
A higher price as a selling point may seem counterintuitive. Why would somebody purchase a product specifically because it costs more, particularly if safer alternatives are available? In a piece for the Wall Street Journal, writer Katherine Sharpe argues that antidepressants are overprescribed in the United States, and discusses the challenges that face a growing segment of population – young people who have no conception of who they are when they aren’t on antidepressants.
In a piece for the Wall Street Journal, writer Katherine Sharpe argues that antidepressants are overprescribed in the United States, and discusses the challenges that face a growing segment of population – young people who have no conception of who they are when they aren’t on antidepressants.