The Mayo Clinic provides some guidance for people who are seeing a doctor about potential mesothelioma, a form of cancer that affects the protective lining that covers many of the internal organs of the body called the mesothelium.
Mesothelioma is caused primarily by exposure to asbestos.
If you have lung or abdominal symptoms that may indicate mesothelioma, according to the Clinic, you’re likely to start by seeing your family doctor or a general practitioner. But when you call to set up an ...
continue reading...
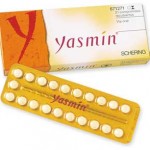 Pharmaceutical giant Bayer is facing legal action from thousands of plaintiffs who claim they or their loved ones have been injured by the
Pharmaceutical giant Bayer is facing legal action from thousands of plaintiffs who claim they or their loved ones have been injured by the  According to a Consumer Reports blog entry, the recent historic $3 billion settlement against pharmaceutical giant GlaxoSmithKline carries some important lessons for consumers.
According to a Consumer Reports blog entry, the recent historic $3 billion settlement against pharmaceutical giant GlaxoSmithKline carries some important lessons for consumers. According to a report on TheHeart.org, the U.S. Food and Drug Administration (FDA) has approved a new vena cava filter made by Crux Biomedical of California.
According to a report on TheHeart.org, the U.S. Food and Drug Administration (FDA) has approved a new vena cava filter made by Crux Biomedical of California.