Specifically, the story focuses on an alleged “phantom recall” of painkiller Motrin. According to the story, the U.S. Food and Drug Administration eventually initiated a nationwide recall of more than 88,000 Motrin tablets because of a problem with the way the drug dissolved.
But the company allegedly contracted workers to buy up Motrin from stores in order to hide the problem and avoid a recall.
In recent years, Johnson & Johnson has faced similar allegations of attempts to conceal defects in its products.
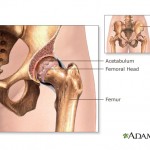
For example, plaintiffs in thousands of lawsuits claim the company sold its all-metal hip replacements and vaginal mesh implants despite being aware of the fact that they could cause debilitating injuries for patients.
Hip implants manufactured by Johnson & Johnson division DePuy Orthopaedics were recalled in 2010 after studied showed that nearly half failed within six years.
And last year, Johnson & Johnson stopped selling four different types of transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, following widespread complaints that they caused severe pain and injuries for recipients.
According to CNN, the FDA classified the Motrin recall as Class 2, which is for products have a low chance of causing major injuries or death, but still pose a risk of “adverse events.”
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip or transvaginal mesh implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://money.cnn.com/2010/06/02/news/companies/mcneil_motrin_phantom_recall/index.htm
