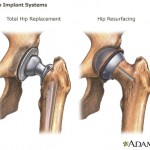
A metal-on-metal implant features both a ball and a cup coated with a combination of cobalt and chromium. The FDA says the metal components slide against each other when the recipient is walking or running, and tiny metal particles may break off – getting into the soft tissues or perhaps even into the bloodstream.
In 2010, metal-on-metal hip implants manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics were recalled because of complaints about metal particles breaking off. Studies also indicated that nearly half of the implants broke down and needed replacement within six years.
The FDA says patients who have received one of the metal-on-metal implants should regard any of the following symptoms as warning signs that the device aren’t functioning properly:
- Pain in the groin, hip or leg
- Swelling at or near the hip joint
- A limp or change in walking ability
If you’ve received a metal-on-metal hip implant, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries, you should also consult with a lawyer familiar with the devices to discuss your legal rights.
See more information here:
