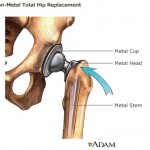
Posing as representatives of a fictitious Chinese company, the reporters presented plans for an all-metal artificial hip joint. Those plans were overtly based on a DePuy Orthopaedics model that has been recalled throughout the world because of safety problems.
Rather than flagging the proposed hip implant as potentially dangerous, however, EVPU officials illegally touted their willingness to actively work for its approval.
The DePuy hip, manufactured by a subsidiary of Johnson & Johnson, was recalled in the United States in 2010. Nearly half of the implants designed along that model, which had both a ball and socket coated in metal, broke down within six years. They also had a tendency to shed toxic metal debris in patients’ bodies.
According to the Daily Mail article, EVPU officials described their approach for gaining approval as a “very fast and easy, easy process,” saying that the hip replacements could be licensed without clinical testing based on their similarity to previously approved devices.
The U.S. Food and Drug Administration approved DePuy hip implants without clinical testing under a similar regulatory loophole.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip implant. If you have significant injuries, you should also consult with a DePuy hip lawyer to discuss your legal rights.
See the story here:
