The report quotes Andrew Boulton, MD, president of the European Association for the Study of Diabetes, as saying: “We feel the proposals are a step in the right direction but we need many more steps to address patient safety.”
The EU approval process for medical devices has recently come under scrutiny following some high-profile device failures. Most notably, a now-defunct French company sold hundreds of thousands of breast implants, which turned out to be prone to rupture and leaking.
Similar high-profile medical device failures in the United States have led to calls for domestic regulatory reforms. Currently, a loophole in the U.S. Food and Drug Administration approval process allows for devices to be approved based on their alleged similarity to previously approved products.
Two devices approved under that loophole have caused thousands of reported injuries for patients who received them.
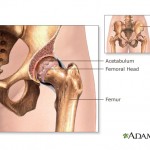
Hip implants manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics were recalled in 2010 after studies showed nearly half of the devices failed within six years, requiring additional surgery for recipients.
And transvaginal mesh implants, used to treat pelvic organ prolapse and urinary incontinence, have generated widespread reports of injury for patients, including infection, chronic pain and organ perforation.
Although the proposed guidelines the EU is considering would widen the reach of devices that are regulated and would empower local authorities to perform more rigorous testing, Boulton and his colleagues in the European diabetes community said the proposal lacks teeth because it doesn’t establish a centralized agency, such as the one that governs medications.
Currently, device makers in EU countries can take their product to any one of more than 70 certified bodies, some of which aren’t even in the EU, for approval.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip or transvaginal mesh implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.medpagetoday.com/MeetingCoverage/EASD/35119
