The proposals come in the wake of a global scandal in which hundreds of thousands of women around the world received breast implants prone to rupture and leaking, made by defunct French company Poly Implant Prothese.
Critics say that because of weak EU safety standards, regulators had failed to stop the implants for more than a decade. Medical devices in Europe are currently overseen by an ad hoc network of up to 80 national assessment agencies.
The new regulations would include establishment of a single panel to monitor the national agencies’ assessments, and would apply to a wide range of products including contact lenses, pacemakers, pregnancy testing kits and high-tech medical equipment such as life support machines.
Consumer safety advocates in the United States have called for a similar overhaul of the country’s approval process, following high-profile medical device failures.
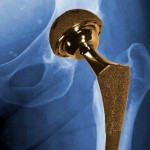
Hip implants manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics were recalled in 2010 after studies showed that nearly half of the devices failed within six years, requiring additional surgery for the patients.
And transvaginal mesh implants, used to treat pelvic organ prolapse and urinary incontinence, have generated thousands of complaints that they failed and caused health problems for patients including infection, chronic pain and organ perforation.
Both devices were approved through a loophole in the U.S. Food and Drug Administration review process, which allows for a device to go on the market based on its alleged similarity to a product that’s already been approved.
In Europe, industry groups are opposing the proposed regulatory overhaul, Reuters says. But consumer advocacy groups say the draft regulations don’t go far enough.
The report quotes Monique Goyens, head of the European Consumer Organization, as saying that the proposed regulations are still less stringent than those applying to medications. And while patients can simply stop taking drugs if there’s a problem, removal of implanted devices may require invasive and risky surgery.
“It is unacceptable that consumers are afforded different protection levels depending whether they have an artificial heart valve or take medicine for diabetes,” Goyens says.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip or transvaginal mesh implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://www.reuters.com/article/2012/09/26/us-eu-medical-devices-idUSBRE88P0OO20120926
