The letter was sent following an agency inspection of the company’s plant in Puerto Rico, which found manufacturing and testing issues related to the company’s Trilogy Acetabular System products — implanted in hip-replacement surgery.
The report came out during a bad news week for manufacturers of hip replacement products.
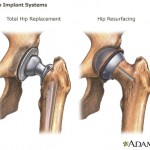
Also this week, a report in the Lancet medical journal recommended that women and smaller men should not get a hip replacement technique called resurfacing, which involves coating both the ball and socket of hip joints with a cobalt and chromium alloy.
U.K. researchers concluded that metal resurfacing implants fail more quickly than total hip replacements made from other materials, and the failure rate is as much as five times higher for women.
Previous research drawing on information from the National Joint Registry for England and Wales fund that all-metal hip resurfacing made by Johnson & Johnson’s DePuy Orthopaedics Inc. division had an even higher failure rate — needing removal or replacement 29 percent of the time after six years, compared with a 9.5 percent failure rate for all metal- on-metal implants.
In the warning letter, the FDA noted that Zimmer hadn’t implemented a testing mechanism to show that its products met design specifications. The FDA also said the company hadn’t validated the effect of certain manufacturing operations on Trilogy products incorporating metallic spikes, which are used to connect the hip replacement cup to a patient’s bone when cement and screws are deemed insufficient.
According to the report, the FDA requires manufacturers of medical products to take steps ensuring the quality of the design and manufacture of its products, known as process validation.
Although Zimmer is notifying customers of the manufacturing issue, the company hasn’t withdrawn any products.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a DePuy hip implant. If you have significant injuries from one of the devices, you should also consult with a DePuy hip lawyer to discuss your legal rights.
See the story here:
https://www.foxbusiness.com/news/2012/10/01/zimmer-receives-fda-warning-on-certain-hip-implants/
