The priority review status is granted by the FDA to expedite the review of drugs to treat serious diseases and fill unmet medical needs. It means the drug will be reviewed in six months rather than the usual 10 to 12-months.
An expedited FDA review process has backfired for Johnson & Johnson in the past.
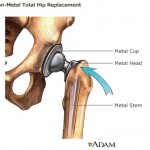
The FDA’s 510(k) process allows a medical device to automatically get approval if it’s deemed “substantially equivalent” to a device already on the market. That regulatory loophole was used to approve “metal-on-metal” hip implants, featuring both a ball and socket coated in a cobalt and chromium alloy, and a type of vaginal mesh implant used to treat urinary incontinence and pelvic organ prolapse.
An all-metal hip implant manufactured by Johnson & Johnson subsidiary DePuy Orthopaedics was subsequently recalled in 2010 following complaints that nearly half failed within six years and required patients to get further surgery. Complaints also included metal debris breaking off and getting into patients’ bloodstream.
Johnson & Johnson also recently recalled four different types of transvaginal mesh implants after hundreds women who had the devices implanted filed lawsuits alleging that they caused severe pain and injury including organ perforation.
The company is trying to get Zytiga approved to treat prostate cancer patients who haven’t undergone chemotherapy. The FDA approved the drug in 2011 to treat prostate cancer patients who have previously received chemotherapy.
Its worldwide sales rose about $32 million sequentially to $232 million in the second quarter, Reuters reports.
You should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries concerning DePuy hips or transvaginal mesh, you should also consult with a lawyer familiar with these cases discuss your legal rights.
See the story here:
https://in.reuters.com/article/2012/08/29/jnj-zytiga-fda-idINL4E8JT54S20120829
