The agency used “spy software” designed to help employers monitor workers, and captured screen images from the government laptops of scientists as they were being used at work or at home.
Though federal agencies have broad discretion to monitor their employees’ computer use, the F.D.A. program may have crossed legal lines by grabbing and analyzing confidential information that is specifically protected under the law, including attorney-client communications, whistle-blower complaints to Congress and workplace grievances filed, the New York Times reports.
The report comes at a time when the FDA is attracting a good deal of criticism.
A number of lawmakers and public health advocates are campaigning for an overhaul of the FDA’s review process for medical devices, which currently allows for medical devices to be approved without clinical testing, based on their alleged similarity to other devices already on the market – even if those original devices were subsequently recalled for safety reasons.
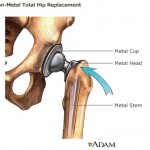
That loophole allowed for the approval of DePuy Orthopaedics all-metal hip implants, which have since been recalled because of their high early failure rate and tendency to shed toxic metal debris in patients’ bodies.
Gynecare Prolift transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, were also approved under that loophole. Thousands of reports have since surfaced of the devices eroding, causing chronic pain, injury and death in patients who had them implanted.
According to the New York Times, the F.D.A.’s spying on its scientists began as a narrow investigation into the possible leaking of confidential agency information, but quickly grew into a much broader campaign to counter outside critics of the agency’s medical review process.
The agency sought to quell what one memorandum called the “collaboration” of the F.D.A.’s opponents, and identified 21 agency employees, Congressional officials, outside medical researchers and journalists thought to be working together to put out negative and “defamatory” information about the agency.
The New York Times says the surveillance effort grew out of a bitter, years-long dispute between the scientists and their bosses at the F.D.A. over the scientists’ claims that faulty review procedures led to the approval of medical imaging devices for mammograms and colonoscopies that exposed patients to dangerous levels of radiation.
A confidential government review in May by the Office of Special Counsel, which deals with the grievances of government workers, found that the scientists’ medical claims were valid enough to warrant a full investigation into what it termed “a substantial and specific danger to public safety.”
You should consult with a doctor if you have any ongoing symptoms or health concerns from a transvaginal mesh or metal-on-metal hip implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
