A Wall Street Journal report on the policy says that it would require the high-risk devices to carry identification numbers. Jeffrey Shuren, director of the FDA’s medical-device center, described the plan as “a major game-changer” and said the agency plans to ramp up efforts to identify malfunctioning medical devices early.
For a long time, public safety advocates have called for a medical device tracking system in the United States. According to the Consumers Union, the lack of such a system has resulted in thousands of patients receiving faulty devices that endanger their health, such as transvaginal mesh and metal-on-metal hip implants.
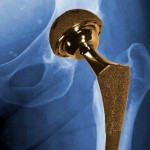
Transvaginal mesh implants, meant to treat pelvic organ prolapse, have turned out to cause problems including organ perforation and debilitating pain for many patients who receive them. And metal-on-metal hip implants have become notorious in recent years both for their high early failure rate and their tendency to leave toxic metal debris in patients’ bodies.
Despite those well-documented problems, both devices are still on the market.
The Consumers Union has pointed out that Australia, England and Wales all maintain lists of every artificial joint implanted, allowing regulators in those countries to identify serious problems with the faulty hip implants years before their counterparts in the U.S.
The new FDA policy calls for the identification numbers to appear on medical devices by early 2014.
According to the Wall Street Journal, some doctors don’t believe the measure will be enough to guarantee patient safety.
The article quotes Robert G. Hauser, a cardiologist with the Minneapolis Heart Institute, as saying the country needs mandatory reporting of device malfunctions.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a transvaginal mesh or metal-on-metal hip implant. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the story here:
https://online.wsj.com/article/SB10001424052702304211804577505094143301240.html?mod=googlenews_wsj
