According to a Bloomberg report, Johnson & Johnson’s prescription drugs and consumer health segments have both suffered revenue losses in recent years following a series of recalls.
Johnson & Johnson has launched about two dozen recalls of nonprescription drugs and at least two of prescription drugs since 2009. In addition, the company has recalled about 25 medical devices and consumer products.
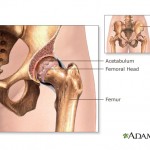
Johnson & Johnson subsidiary DePuy Orthopaedics recalled a line of all-metal artificial hip implants in 2010 because nearly half of all recipients had to get a replacement within six years, and many also suffered metal toxic metal debris breaking off in their bodies.
And the company recently announced that it will stop selling four types of vaginal mesh implants used to treat urinary incontinence and pelvic organ prolapse, after more than 600 women filed lawsuits claiming the devices caused injuries and debilitating pain.
Johnson & Johnson hopes that Synthes will generate business in emerging markets such as Russia, China and India — where patients may get bones and joints fixed after accidents but rarely can afford expensive elective surgeries such as hip and knee replacements.
The report says orthopedics treatments in general are a fast-growing area, given the aging worldwide population and the wear and tear that obesity puts on bones. Rising government health spending and a growing middle class in emerging markets are also fueling growth, according to Bloomberg.
You should consult with a doctor if you have any ongoing symptoms or health concerns from a Johnson & Johnson product. If you have significant injuries, you should also consult with a DePuy hip or transvaginal mesh lawyer to discuss your legal rights.
See the report here:
https://www.businessweek.com/ap/2012-06/D9VBST9G4.htm
