At issue is whether the company misled Medicaid officials about the risks of antipsychotic drug Risperdal and marketed it for unapproved uses, duping the state into improperly paying millions more for the drug than it should have.
According to an article in the Philadelphia Inquirer, a lawyer for Johnson & Johnson argued that Pennsylvania failed to prove its case. But a lawyer for the state produced two letters from the U.S. Food and Drug Administration criticizing a Johnson & Johnson subsidiary’s promotional materials on Risperdal as false and misleading.
The Commonwealth Court is considering an appeal of a Philadelphia judge’s 2010 decision to throw out a previous state lawsuit against Johnson & Johnson.
Allegations against Johnson & Johnson in other states have resulted in big payouts from the company.
A judge in Arkansas fined Johnson & Johnson $1.2 billion after a jury ruled that the company had, in fact, used deceptive tactics to market Risperdal. A total of 11 states have filed similar lawsuits.
The company is also facing legal difficulties over allegations that it improperly marketed dangerous medical devices.
Up to 10 percent of patients who received transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, had the devices fail within a year. The most common reported problem is the vaginal mesh eroding and sticking through the walls of the bladder and vagina, causing debilitating pain for patients. The company is still marketing the device, despite thousands of such cases.
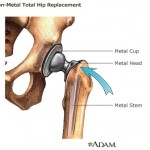
And the U.S. Food and Drug Administration is investigating metal-on-metal hip implants manufactured by a subsidiary of Johnson & Johnson in response to thousands of reports that the devices fail prematurely and leave toxic metal debris in patients’ bodies.
The hip implants were recalled in 2010. But an investigation by British medical journal The Lancet found internal company documents indicating that Johnson & Johnson officials were aware as early as 2005 of the device’s high early failure rate, yet continued to market it.
If you’ve received either of these devices, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries, you should also consult with a lawyer familiar with the DePuy hip implant or transvaginal mesh case to discuss your legal rights.
See the Inquirer story here: https://www.philly.com/philly/business/151807935.html
