According to an article in the Philadelphia Inquirer, Pennsylvania previously filed a lawsuit alleging that Johnson & Johnson tricked the state into paying millions more for the drug than it should have. But a Philadelphia judge threw it out in 2010.
A jury in Arkansas recently ruled that Johnson & Johnson used deceptive tactics to market antipsychotic drug Risperdal, misleading doctors about the drug’s risks and improperly marketing it for unapproved uses. A judge fined the company $1.2 billion.
That marked the third such jury verdict finding that Johnson & Johnson concealed Risperdal’s risks and tricked Medicaid regulators into paying more than they should have for the medication. A total of 11 states have sued Johnson & Johnson in an attempt to get reimbursement for Medicaid or other public funds paid on Risperdal prescriptions.
According to the Inquirer, the Risperdal litigation stemmed from an investigation that Allen Jones, a former investigator for the state Office of Inspector General, began in 2002. Johnson & Johnson was allegedly funneling money to Pennsylvania health officials who helped develop state medication policy.
Those aren’t the only allegations of unethical behavior leveled at the company.
Plaintiffs in thousands of cases against Johnson & Johnson claim the company used similar deceptive tactics in marketing two medical devices.
Up to 10 percent of patients who received transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, had the devices fail within a year. Those patients were left to deal with medical problems including organ perforation and debilitating pain.
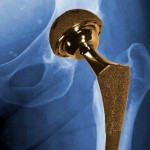
And the U.S. Food and Drug Administration is investigating metal-on-metal hip implants manufactured by a subsidiary of Johnson & Johnson in response to thousands of reports that the devices fail prematurely and leave toxic metal debris in patients’ bodies.
If you’ve received either of these devices, you should consult with a doctor if you have any ongoing symptoms or health concerns. If you have significant injuries, you should also consult with a lawyer familiar with the DePuy hip implant or transvaginal mesh case to discuss your legal rights.
See the Inquirer story here: https://www.philly.com/philly/business/151656315.html
