That 12.5-percent increase came about primarily because of lower spending on research, sales and administration and a boost from selling rights to a drug, the report said.
According to the report, Johnson & Johnson continues to lose hundreds of millions of dollars a year in revenue because of problems related to manufacturing quality.
Another recent setback for Johnson & Johnson was an Arkansas jury’s ruling that the company used deceptive tactics to market antipsychotic drug Risperdal, which included misleading doctors about the drug’s risks and improperly touting it for unapproved uses.
A total of 11 states have sued Johnson & Johnson, alleging that the deceptive marketing led to them overpay from Medicaid and other taxpayer-supported funds.
Johnson & Johnson has been accused of similar deceptive tactics in marketing two medical devices.
Transvaginal mesh implants, used to treat urinary incontinence and pelvic organ prolapse, have been failing within a year for up to 10 percent of the patients who received them, according to one study, leaving patients with problems including organ perforation and debilitating pain.
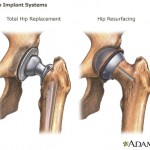
And metal-on-metal hip implants manufactured by a subsidiary of Johnson & Johnson are being investigated by the U.S. Food and Drug Administration after thousands of reports that they fail prematurely and leave toxic metal debris in patients’ bodies.
Plaintiffs in thousands of lawsuits centering on both devices claim the company actively marketed them despite being aware of those problems.
See the report here: https://www.businessweek.com/ap/2012-04/D9U6QEDG0.htm
